News: New Electrodes Turn Normal Batteries Into Batteries That Recharge in a Flash
May 12, 2011- Saber Miresmailli
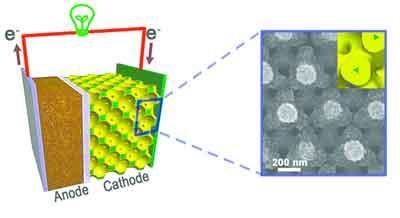
Left: diagram of a lithium-ion battery constructed using a nanostructured bicontinuous cathode. Right: scanning electron microscope image of the nanostructure, a three-dimensional metal foam current collector coated with a thin layer of active material. Image courtesy of Paul Braun and Huigang Zhang, University of Illinois.
Imagine a day when you can charge your cellphone in 30 seconds, your laptop in 2 minutes and your electric car in 5 minutes. Thanks to researchers at the University of Illinois and their new invention, that day can be as early as two years from now.
Professor Paul Braun, Dr. Huigang Zhang and Dr. Xindi Yu at the Department of Material Science and Engineering developed a three-dimensional nanostructure for battery cathodes that allows for dramatically faster charging and discharging without sacrificing energy storage capacity. The researchers' findings were published in the March 20 advance online edition of the journal Nature Nanotechnology.
This has been achieved by using a self-assembling three-dimensional nanostructure for battery cathodes in which tiny spheres are packed onto a surface and form automatically into a lattice. Once in place, metal is packed around the spheres which are then dissolved, leaving a sponge-like 3-D metal scaffolding. The spaces within the scaffolding are enlarged using an “electropolishing” technique and then filled with a thin-film active material.
"This system that we have gives you capacitor-like power with battery-like energy," said Prof. Braun. "Most capacitors store very little energy. They can release it very fast, but they can't hold much. Most batteries store a reasonably large amount of energy, but they can't provide or receive energy rapidly. This does both. By changing the nanostructure of the electrodes, we provide a pathway for ions so what it normally take minutes to hour can now be done in seconds.”
This technology can dramatically enhance the convenience of electric cars. “If you had the ability to charge rapidly, instead of taking hours to charge the vehicle you could potentially have vehicles that would charge in similar times as needed to refuel a car with gasoline,” said Prof. Braun. “If you had five-minute charge capability, you would think of this the same way you do an internal combustion engine. You would just pull up to a charging station and fill up.”
All type of batteries can benefit from this new technology. "We like that it's very universal, so if someone comes up with a better battery chemistry, this concept applies," said Braun, who is also affiliated with the Materials Research Laboratory and the Beckman Institute for Advanced Science and Technology at Illinois. "This is not linked to one very specific kind of battery, but rather it's a new paradigm in thinking about a battery in three dimensions for enhancing properties."
This new technology will not change the way we recycle the batteries. “We used nickel or aluminum for the scaffold and both of them are easy to recycle. There is nothing uniquely toxic in this technology. These batteries should recycle just like any other batteries. They are no better and they are no worst” Said Prof. Braun. More over this technology can improve the energy sustainability of current batteries. “In normal batteries that charge or discharge over long periods of time, energy will be loosed in form of heat. This technology enable us to minimize the energy loss” said Dr. Zhang, the co-inventor of the 3D electrodes.
This project was sponsored by U.S. Army Research Laboratory and the Department of Energy.
Nature Nanotechnology, 2011. DOI: 10.1038/NNANO.2011.38 (About DOIs).
Category: